Section: 300-3
Effective: 07/01/1991
Supersedes: 01/05/1976 350-3
Next Review Date: TBD
Issuance Date: 07/01/1991
Issuing Office: Extramural Funds
NON-PAYROLL EXPENSE TRANSFERS
I. REFERENCES AND RELATED POLICIES
A. Systemwide Business and Finance Bulletin (BFB)
A-47 University Direct Costing Procedures
B. UCSD Policy and Procedure Manual (PPM)
C. Office of Management and Budget Circular No. A-21
D. Public Health Service Grants Policy Statement
E. Contract and Grants Operating Guidance Memos: No. 90-14 and No. 90-16
F. Federal Demonstration Project General Terms and Conditions
II. BACKGROUND
This policy establishes guidelines for transferring expense (or direct cost) either after it has been recorded in the ledgers by an authorized transaction for an initial payment or by a recharge from a University administrative or service unit from one accounting distribution to another. Within the context of this policy and procedure, a department refers to an administrative unit, office, project, or activity which is identified by a unique organization/program/fund designation.
A "FDP transfer" refers to a transfer between two or more Federal Demonstration Project (FDP) awards. A non-FDP transfer refers to a transfer between a Federal award (FDP or non-FDP) and any other fund source, extramural or non-extramural. Transfers between FDP and non-FDP awards are cited in regulations and policy entered under I. References.
III. POLICY
A. Authorized Transfers
Policy provides that original certification as to the appropriateness of an expenditure has been accepted as being correct. It is appropriate to make an expenditure adjustment from one accounting distribution to another only in the following situations:
1. For All Funds
a. To correct an erroneous recording, such as when the original source document(s) cited an incorrect accounting distribution.
b. To record a change in the original determination as to the use of goods or services (for example, a case of beakers originally ordered for and charged to a teaching program can be subsequently transferred to a research project).
2. Federal and Private Contract and Grant Awards
When closely related work is supported by more than one funding source, a transfer of expense from the originally charged funding source to another can be made provided that the request meets all of the following:
a. The cost is a proper and allowable charge to the new fund source.
b. The request is supported by documentation which contains a complete explanation and justification for the transfer including the method of proration.
3. FDP Awards
When the transfer is between two or more FDP awards other than National Institute of Health (NIH) or Alcohol, Drug Abuse and Mental Health Administration (ADAMHA), the costs can be charged or transferred to either or any of the awards involved as long as the productivity and the approved scope of those awards are maintained and the costs are otherwise allowable.
For NIH and ADAMHA awards, the benefit must be established, if possible, and the cost allocated based on proportional benefit. "However", when a cost benefits two or more projects or activities in proportions that cannot be determined because of the interrelationship of the work involved, the cost may be allocated to the benefitted projects on any reasonable basis.
B. Standards For Transfers: Federal Contracts and Grants
A request for the transfer of an expenditure to a Federal contract or grant must meet the following criteria:
1. Allowable Costs
Costs must conform to any limitations or exclusions set forth in "OMB Circular A-
21 Cost Principles" (see I.C.); the award document, or State and Federal regulations.
2. Reasonable Costs
Costs may be considered reasonable if they reflect the action a prudent person would have taken under similar circumstances, are of a type considered necessary, and reflect the constraints of arm's-length bargaining.
3. Allocable Costs
A cost is allocable to a particular award, if it can be assigned with a high degree of accuracy in accordance with relative benefits received or other equitable relationship.
4. Consistent Treatment
Costs should be treated consistently under similar circumstances in accordance with generally accepted accounting practices.
5. Reasons of Convenience
For non-FDP awards, costs may not be shifted in order to meet deficiencies caused by overruns or other fund considerations. For FDP awards see III.A.3. above.
This policy will be applied equally to all non-payroll and payroll expenditure transfers on the San Diego Campus (see PPM 395-4.3, Timekeeping: Review of Charges for additional policy and procedure).
IV. PROCEDURE
To initiate a non-payroll expense transfer, a Non-Payroll Expenditure Transfer (NPET), Form 2047, Exhibit A, must be completed by the Department.
A. Operating Ledger Information
The following information from the operating ledger must be entered onto the form:
1. Accounting Information: Organization name, Index, Fund, and Account number.
2. Document No.: Ledger column titled "Document"
3. Reference: Ledger column titled "Reference"
4. Ledger Date: Date of Operating Ledger
5. Totals: The totals for the "Debit" and "Credit" columns must be of equal amounts and must be entered on the "Totals: line of the form. The sum total of the debit and credit amounts must be entered on the "Document Total" line of the form.
6. Ledger Amount: The expense must be in the same amount originally recorded in the operating ledger (i.e., the whole amount or the appropriate fraction charged to and/or paid by the unit requesting the adjustment).
B. Justification
1. Explanation
a. Non-FDP Transfers
A detailed explanation for each transfer of expense must be entered in the "Explanation" section of the form. An explanation which merely states that the transfer is "to correct error" or "to transfer to correct project" is insufficient.
b. FDP Transfers
The explanation need only state: "To transfer costs to the FDP award(s) designated by the Principal Investigator for allocation of these expenditures." Transfers to NIH and ADAMHA grants will either have to indicate the benefit received or state that "The benefit cannot be readily determined and the Principal Investigator is allocating costs based on his/her best judgement."
c. Gift Funds and Non-Extramural Fund Sources
A brief explanation for each transfer of expense must be entered in the
"Explanation" section of the form.
2. Timeliness
To the maximum extent possible, an expense transfer should be made within 120 days of the original charge. Any attempt to transfer cost more than 120 days after the original charge, raises serious questions concerning the propriety of the transfer. Therefore, if a transfer is necessary after the 120 day period, the supporting documentation, in addition to the required explanation, must also include a detailed explanation as to the untimeliness of the transfer or the transfer will not be processed. This also applies to FDP transfers.
C. Documentation
1. Non-FDP Transfers
Supporting documentation should include a copy of the Purchase Order, Purchase Requisition, Invoice, or Recharge billing form, whichever is applicable, and a copy of the Operating Ledger showing the original expense. All copies should be annotated to reflect the correct index or fund number.
2. FDP Transfers, Gift Funds and Non-Extramural Fund Sources
The ONLY documentation required is a copy of the Operating Ledger (annotated to reflect correct index code).
D. Certification
The Non-Payroll Expense Transfer (NPET) must be properly certified and authorized on the lines marked "Department Approval:."
1. For All Extramural Awards
When an expenditure is adjusted between departments or within the same department between accounting distributions with different signature authorizations, the appropriate authorized signature for each accounting distribution must be provided. Also the signatures in the "Prepared By:" and "Department Approval:" section MUST be by two different individuals.
2. Non-FDP Transfers
For direct charges to Federal contracts and grants, PI or Department Chair
MUST sign the transfer.
3. FDP Transfers
Signature authorization for Principal Investigators may be delegated to the position of administrative analyst or higher. If authorized signature is someone other than the Principal Investigator, a memo signed by the Principal Investigator delegating signature authorization to individual signee must be maintained in the department's files.
E. Processing
The originating department forwards both copies (white original and yellow duplicate) of the Non-Payroll Expenditure Transfer (NPET) form to the Accounting Office,
Mail Code 0954.
V. RESPONSIBILITIES
A. Accounting Office
1. Approves the forms after ensuring that the adjustments are in compliance with applicable campus, University, and Federal policies.
2. Assigns "document numbers" to the forms and returns the yellow duplicate copies to the originating department.
3. As the Office of Record, enters the forms into the financial data base and files the original NPET in compliance with record retention requirements.
B. Principal Investigator
Ensures that all charges to Federal contracts and grants, and State and Private grants and agreements are appropriate, accurate and in accordance with agency requirements.
C. Departments
Ensure that NPET forms are complete and accurately contain all pertinent information and support documentation as cited in this PPM.
EXHIBIT A Journal Type: Document #:
UCSD NON-PAYROLL EXPENDITURE TRANSFER
University of California, San Diego (Preparation Instructions on next page)
Effective Date / / Page of
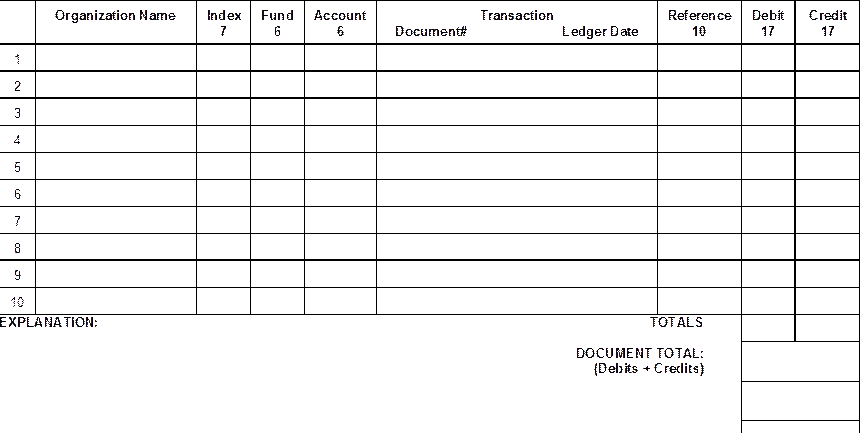
Prepared by:
Accounting Office Audit:
Phone Mail Code Date Date
Departmental Approval:
Accounting Office Approval:
Date Date
Departmental Approval:
Date
FO2047(7/91) Distribution: White--Accounting Yellow--Return to Department by Accounting Retention: 10 Years subject to Contract and Grant Requirements
NON-PAYROLL
EXPENDITURE TRANSFER
INSTRUCTIONS
1. Use this form to transfer non-payroll charges between organizations and/or funds by Index code.
2. Each month the Accounting Office provides departments with an Operating Ledger, UGAP100, listing all charges to expenditure accounts. If incorrect recordings of non-payroll expenses are noted, transfers are to be reported by using this form. Each item of information requested on this form is available from the Operating Ledger report.
3. Departmental personnel must fill in the form completely with the information shown in the Operating Ledger report. The form can be either typed or handwritten in ink. It is important that information be entered within the spaces and with no overlapping.
4. A separate line on the form must be used for each line item on the Operating Ledger.
5. The reason for each line item of the expense transfer must be given for audit purposes.
A. FDP Transfers (between two Federal Demonstration Project awards)
1. Include this statement: "To transfer costs to the FDP award(s) designated by the Principal Investigator (PI) for allocation of these expenditures." (If and NIH or ADAMHA award state the benefit to the debited award).
2. Explain the untimeliness if past 120 days (four months from the Operating Ledger date).
3. Attach copies of the operating Ledger showing the original expense.
B. Non-FDP Transfers
1. Explain how the error in recording occurred.
2. Explain the benefit to the debited award.
3. Explain in the untimeliness if past 120 days (four months from the Operating Ledger date).
4. Explain the method of proration (if used) among Index codes.
5. Attach copies of supporting documentation (e.g. Purchase Order, invoice, recharge form, Operating Ledger) annotated to reflect correct Index code.
C. All other Funds
1. Explain how error in recording occurred.
2. Attach copies of supporting documentation annotated to reflect correct Index code.
6. The form must be approved with signatures authorized for Funds/Organizations/Programs of the Index codes cited. Note that the "Prepared by" and "Department Approval" MUST be different signatures for Extramural Funds.
A. FDP Transfers - Signature authorization should be delegated only to the position of Administrative Analyst or higher.
B. Non-FDP Transfers - For charges to Federal contracts and grants, the Principal Investigator or Department Chair MUST sign the form.
7. The original (Accounting Office) and Departmental copy must be sent to the Accounting Office for recording. The Departmental copy will be returned to the department.
8. Refer to Policy and Procedure Manual chapter 300-3 "Non-Payroll Expense Transfers" for further information.
